// P02
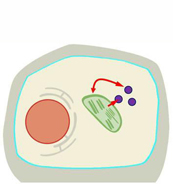
Interplay of plastids and peroxisomes in jasmonate biosynthesis
Project Leader// Prof. Dr. Bettina Hause
Leibniz Institute of Plant Biochemistry
Department of Cell and Metabolic Biology
Jasmonate Function & Mycorrhiza
Jasmonic acid (JA) and its derivatives mediate plants responses to various biotic and abiotic stresses, but are also regulators of plant development. Jasmonates are synthesized from α-linolenic acid released from plastid membranes. The first three steps of the biosynthesis leading to the intermediate cis-12-oxo-phytodienoic acid (OPDA) are located in plastids. The conversion of OPDA to JA, however, is located in peroxisomes, whereas the final conversion of JA to JA-isoleucine takes place in the cytosol. Since OPDA is synthesized in plastids, its transport from plastids into peroxisomes is required for JA biosynthesis. Therefore, it is likely that the control of its movement between these subcellular compartments is highly regulated. Both, plastids and peroxisomes, are dynamic organelles and are often located in close proximity. This raises the question, whether certain developmental stages of plastids and differently proliferated peroxisomes contribute differently to JA biosynthesis. Therefore, the aim of this project is to answer the question, whether JA biosynthesis is dependent on plastid development and peroxisome proliferation. It is planned to visualize the dynamics of both organelles upon stress treatment leading to synthesis of JA (e.g., wounding) using transgenic plants expressing fluorescent proteins. These analyses require the use of advanced microscopic methods and will allow the selection of tissues exhibiting different dynamics of both organelles, different types of plastids, or differences in the presence, size and proliferation of peroxisomes. The capacity of these tissues to synthesize OPDA and JA will be monitored by LC-MS analysis. In addition, it is aimed to functionally characterize previously identified OPDA-induced genes by gain- and loss-of-function approaches in A. thaliana.


